The Congressionally Directed Medical Research Programs’ (CDMPR), Peer Reviewed Cancer Research Program (PRCRP) consumer advocates Katie Jamieson and Jessica Blackford-Cleeton recently participated in the evaluation…
Today, the Mesothelioma Applied Research Foundation announced that it will fund three research projects in the field of mesothelioma for a total investment of $300,000…
At the end of May, in a brand new virtual format, the annual meeting for the American Society of Clinical Oncology (ASCO) was one of…
A new approach of anti-PD1 immunotherapy (atezolizumab) combined with bevacizumab (an agent that blocks the growth of new blood vessels therefore limiting the supply of…
A phase 2 immunotherapy clinical trial combining durvalumab, an immunotherapy drug, with standard treatment of Alimta (pemetrexed)/cisplatin has shown promising overall survival results. The study enrolled 55 patients and…
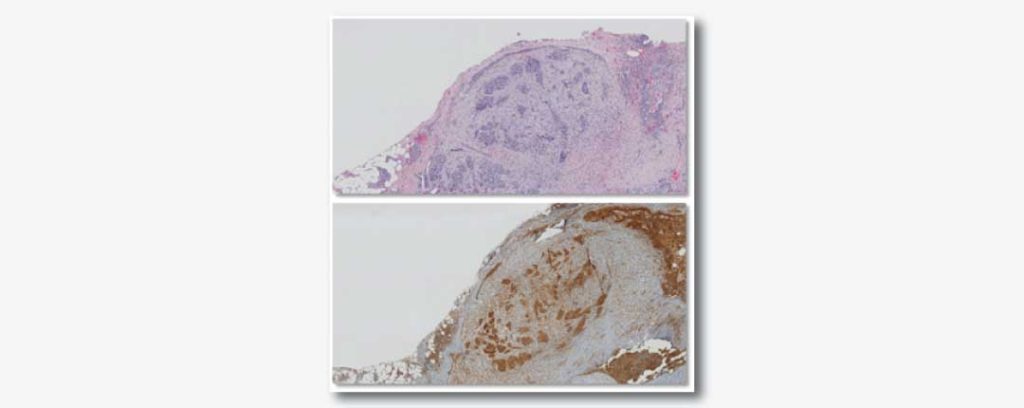
In 2016, the Mesothelioma Applied Research Foundation funded a grant to Antonio Giordano, MD, PhD, director and founder of the Sbarro Health Research Organization at Temple University, to investigate how silencing…